Equity research Immunovia Q1 2026: Initial commercialisation phase on track
11 May 2026
Read the full report here:
The recent quarter confirms that Immunovia is well on track to significantly increase its base of US centres for surveillance of pancreatic cancer risk registered to use the PancreaSure test. Going forward, the company highlights Medicare coverage submissions in Q3 2026 and partnership discussions as key priorities.
Solid pace in penetration of US surveillance centres
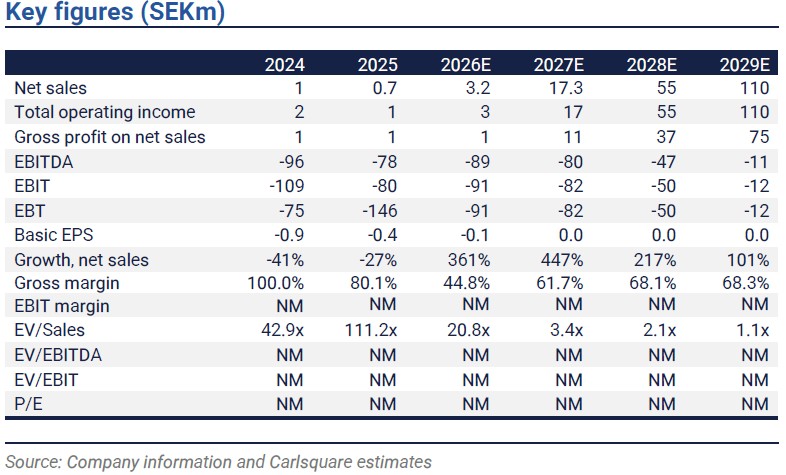
Immunovia has maintained strong momentum in enrolling high-risk surveillance centres (HRSCs) as registered users for the PancreaSure test in Q1 2026, increasing the total to 21 from 12 during the period. Actual net sales amounted to SEK 0.4m (0.1), consisting of both royalties and PancreaSure sales. Total net sales were thus close to our SEK 0.5m forecast. Immunovia reiterates the previously stated target to submit for Medicare coverage in mid-2026. With Medicare and private insurer coverage, the average payment per test may eventually increase to ~4x the current level (before any split with potential future distribution partners). At this point, we expect sales in Q2 2026 to be similar to Q1, followed by a possible pickup towards the end of the year, as Immunovia aims to increase use among its growing base of penetrated centres.
Discussions with potential commercialisation partners a key priority
OPEX was flat and lower than expected, at SEK -19m. Cash burn was below previous company guidance, and Immunovia reiterates that its cash balance will secure its working capital needs through Q3 2026. In the short term, this allows for increased clinical activity, such as the new registry study, ASSURE, which assesses the clinical utility of PancreaSure. The aim is to generate additional evidence supporting the process of establishing reimbursement. By the end of Q1, the cash position was SEK 56m, down from SEK 77m in the previous quarter. There are clear signals that Immunovia is giving business development and partnerships activities higher priority. Management mentions discussions with over a dozen prospective U.S. commercialisation partners. Also, the board is proposing an incentive program tied to certain “exit events” including outlicensing of PancreaSure or significant ownership changes in Immunovia. We believe proven commercial traction for PancreaSure is likely a key parameter for successful partnering on favourable terms.
Reimbursement timelines determine short- and medium-term forecasts
We reduce our short- to medium-term sales estimates somewhat (by 8% on average 2026-2027E), mainly because we now expect average revenue per test to be initially lower than previously assumed in this admittedly early phase of commercialisation. This adjustment is further impacted by an extension of assumed reimbursement timelines, e.g., for Medicare coverage, in our model. For the earnings forecast, the reduced near-term sales expectations are largely mitigated by better financial discipline/lower OPEX than we had previously anticipated. However, the current subdued share price again motivates adjustments to our assumptions about future equity financing and dilution. The result is a moderate tweak of our base-case valuation.
Disclaimer
Carlsquare AB. www.carlsquare.se, hereafter referred to as Carlsquare, conducts operations in Corporate Finance and Equity Research and thereby publishes information about companies, including analyses. The information has been compiled from sources that Carlsquare considers reliable. However, Carlsquare cannot guarantee the accuracy of the information. Nothing written in the analysis should be regarded as a recommendation or invitation to invest in any financial instrument, option or similar. Opinions and conclusions expressed in the analysis are intended solely for the recipient.
The content may not be copied, reproduced, or distributed to any other person without the written consent of Carlsquare. Carlsquare shall not be liable for any direct or indirect damage caused by decisions made based on information contained in this analysis. Investments in financial instruments provide opportunities for capital appreciation and profits. All such investments are also associated with risks. The risks vary between different types of financial instruments and combinations thereof. Historical returns should not be considered as an indication of future returns.
The research is not directed at U.S. Persons (as that term is defined in Regulation S of the United States Securities Act and interpreted in the United States Investment Companies Act 1940) and may not be distributed to such persons. Nor is the analysis aimed at such natural or legal persons where the distribution of the analysis to such persons would involve or entail a risk of violation of Swedish or foreign law or regulations.
The analysis is a so-called commissioned analysis where the analysed company has signed an agreement with Carlsquare for analysis coverage. The analyses are published continuously during the contract period and against customary fixed remuneration.
Carlsquare may or may not have a financial interest in the subject of this analysis. Carlsquare values ensuring objectivity and independence and has therefore established procedures for managing conflicts of interest.
The analyst Niklas Elmhammer does not and may not own shares in the analysed company.







